GOLDMINE Study
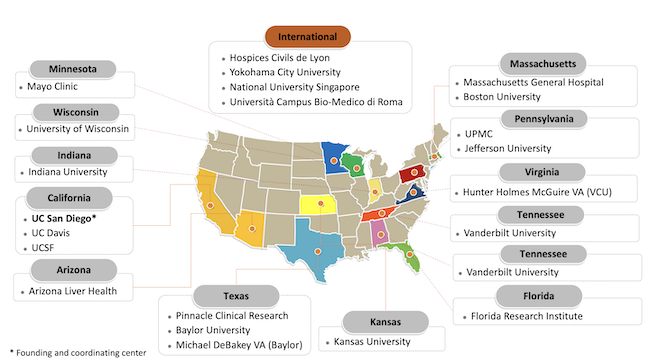
GOLDMINE Study participating sites
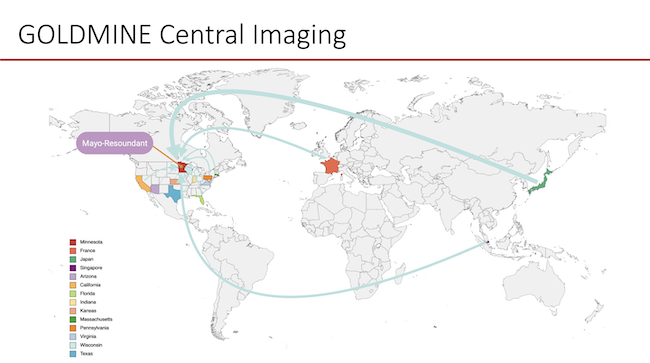
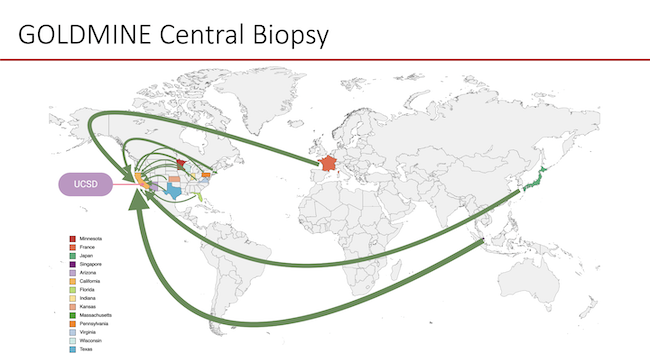
Global Longitudinal Assessment of Nonalcoholic Fatty Liver Disease (MASLD) using Magnetic Resonance Elastography (GOLDMINE) study
The purpose of the GOLDMINE study is to better understand MASLD by yearly following and evaluating people with biopsy-proven or a clinical diagnosis of MASLD. MASLD is a common liver disease in the United States. The causes of MASLD are unknown, but are probably due to different reasons, from inherited characteristics to personal lifestyle. Some reasons may increase the chance of developing MASLD (these are called “risk factors”) while other reasons may decrease the chance of developing MASLD (“protective factors”). The factors that determine whether a patient progresses from MASLD to more severe liver disease are unknown. The time it takes for a patient to progress to more severe disease is also uncertain.
The term MASLD covers a range of liver disease progression. When fatty liver disease moves on to a more severe level of liver injury, it is called metabolic dysfunction-associated steatotic liver disease (MASH). MASLD/MASH is likely the most common liver disease in the United States and is thought to be related to obesity or diabetes. A liver biopsy (a small amount of liver tissue removed by needle) is usually done to confirm the liver disease. The biopsy results may show different amounts of fat, inflammation (swelling), and scarring in the liver. MASH can lead to severe liver disease in some patients.
MASLD is usually discovered because of abnormal liver tests or from a liver ultrasound in persons with normal liver tests. The few known risk factors include high blood sugar and fat levels in the blood. Patients with MASLD often have resistance to the normal action of insulin, a hormone that is important for processing sugar and fat. Increased resistance to insulin can lead to fat in the liver.
The main purpose of this study is to learn more about fatty liver disease by collecting information locally and in partnered sites. This study includes one primary clinic visit and up to 10 additional annual follow-up visits with a minimum of 4 years follow-up requirement.
To be eligible for this study, you must be at least 18 years old at the time of initial screening. We plan to recruit a total sample of 1000 adult participants with biopsy-proven MASLD or known MASLD-related cirrhosis. Approximately, 500 participants will be enrolled at UCSD.
Note: We are currently enrolling patients for this study.
For additional questions regarding this study please contact:
Phone: (858) 246-2179
masld@health.ucsd.edu
